What is TDS?
TDS is Total Dissolved Solids. Water dissolves the minerals present in the strata of soil it filers through in the case of ground water and, in the case of surface water, the minerals present in the soil over which it flows (rivers/streams) or over which it stands (lakes, ponds, reservoirs).The dissolved minerals in water are commonly referred to as Total Dissolved Solids (TDS). The TDS content of any water is expressed in milligrams /litre (mg/l) or in parts per million (ppm). These units are equivalent.
The minerals are basically compounds (salts) of Calcium(Ca), Magnesium(Mg) and Sodium(Na) What is commonly called as ‘hardness in water’ is due to the compounds/salts of Ca and Mg such as Calcium or Magnesium Chloride, Calcium or Magnesium Sulphate ( CaSo4, MgCl, etc).Some types of dissolved solids are specifically dangerous even in low quantities. This includes arsenic, fluorides and nitrates. There are particular standards for the acceptable amounts of these elements in water and in some cases like fluoride; there is some disagreement as to what constitutes safe levels.
Leaving aside the specific harmful chemicals fluoride and arsenic, drinking water for human beings should contain some level of minerals (TDS), but these levels should not be excessive.
What are the TDS Standards?
India Standards: The standard that applies to India is the BIS 10500- 2012 standard
This standard used the WHO standard as the basis and has been amended subsequently to take into account the fact that over exploitation of ground water which has the largest share of water supplied for human use has deteriorated to such an extent that the crucial parameters such as TDS, hardness, Chlorides, etc usually exceed the desirable levels substantially. Consequently, a higher permissible limit has been specified. Water used for drinking becomes unpalatable when the TDS level is above 500 mg/l, but lack of any better source enables people consuming such water to get used to its taste. The BIS standard applies to the purity level acceptable for human beings to drink. For practically all industrial and some commercial uses, the purity levels required are very much higher and in most cases demand water with virtually no residual dissolved solids at all.
BIS Standard says that the maximum desirable TDS is 500 mg/L and the maximum permissible level in the absence of a better source of water is 2000 mg/L. A related standard is the ‘hardness measured as CaCO3″ where the acceptable limit is 200 mg/L and maximum permissible is 600 mg/L.
WHO Standards:
“Water containing TDS concentrations below 1000 mg/litre is usually acceptable to consumers, although acceptability may vary according to circumstances. However, the presence of high levels of TDS in water may be objectionable to consumers owing to the resulting taste and to excessive scaling in water pipes, heaters, boilers, and household appliances (see also the section on Hardness).
Water with extremely low concentrations of TDS may also be unacceptable to consumers because of its flat, insipid taste; it is also often corrosive to water-supply systems “
Reference: http://www.who.int/water_sanitation_health/dwq/chemicals/tds.pdf
US EPA Standard:
The U.S. Environmental Protection Agency (EPA) recognises broadly two categories of drinking water standards, known as maximum-contaminant-level goal (MCLG) and secondary maximum contaminant level (SMCL). The MCLG is a health goal set at a concentration at which no adverse health effects are expected to occur and the margins of safety are judged “adequate,” while the SMCL is a non-enforceable guideline that presents no risk to human health. While fixing no limit for MCLG, the EPA has fixed an upper limit of 500 mg/L for SMCL. This limit has been fixed to avoid undesirable aesthetic effects of odour, taste and colour that could be felt by consumers and technical effects of corrosion, incrustation, staining, scaling and sedimentation of pipelines and other fixtures that convey water. Despite not fixing a limit to MCLG of TDS, high TDS water can have certain other constituents at harmful levels of SMCL to cause adverse health effects. Thus MCLG can be a few times more than the SMCL.
Very low TDS: Due to insipid or bitter taste and lack of useful minerals, too-low TDS also causes problems. There does not seem to be a generally accepted lower limit.
How to measure TDS?
The standard for monitoring the purity of water by electrical resistance is termed specific resistance corrected to 25°C or R-25. Specific resistance for this purpose is based on the resistance of an electrical current between two 1 cm square plates spaced 1 cm apart as measured at 25°C. The space between the plates is a 1 cm cube.
The resistivity of absolute pure water is 18.2 (rounded) MΩ × cm at 25°C or 0.055 micro-siemens/cm. Water of this quality must be measured inline (closed system) in order to prevent atmospheric interference of the reading. As water is drawn from a water purification system that is showing 18.2 MΩ × cm purity, carbon dioxide from the atmosphere is immediately absorbed into the solution. The carbon dioxide reacts with water forming carbonic acid in solution.
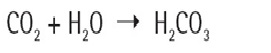
Carbonic acid disassociates in water forming counter ions, which conduct electrical current.
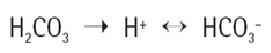
This will drop the specific resistance of the water to below 8 or 10 MΩ × cm in less than a minute.
As previously mentioned, a specific resistance 18.2 MΩ × cm (million ohms) at 25°C is considered to be absolute pure water. This only accounts for the dissolved ionic impurities commonly found in water. Organic materials found in water cannot be directly detected by resistivity | conductivity. Total organic carbon (TOC) analysis or a chromatographic method is needed to screen water for this type of generic or specific contaminant.
Natural or municipally treated waters will contain an infinite range of TDS. Some water sources may have a TDS below 50.0 ppm or over 800.0-ppm. The type of dissolved material found in a water supply may also vary. Typically, potable waters will contain a certain amount of calcium, magnesium and sodium with counter ions such as carbonates, sulfate and chloride. These materials originate from water contact with rocks and minerals found in the Earth’s crust. As water passes through the crust, these materials are dissolved and carried into rivers, lakes and reservoirs used for potable water distribution. Simply stated, sodium chloride (NaCl table salt) will dissolve in water to form disassociated ions.

The same will happen with the other mineral salts as they dissolve. These mineral salts provide the means for water to conduct an electric current. Therefore, specific resistance or conductance can be used to estimate the amount of TDS in a given water supply. It should be noted that TDS might fluctuate considerably from any source. For example, a water sample having a specific resistance of 4000 Ω × cm would contain about 125.0 ppm of TDS. A sample with a 600 Ω × cm specific resistance would have TDS of about 835.0 ppm.
In Conclusion:
In general, the total dissolved solids concentration is the sum of the cations (positively charged) and anions (negatively charged) ions in the water. Therefore, the total dissolved solids test provides a qualitative measure of the amount of dissolved ions but does not tell us the nature or ion relationships. In addition, the test does not provide us insight into the specific water quality issues, such as Elevated Hardness, Salty Taste, or Corrosiveness. Therefore, the TDS (total dissolved solids) test is used as an indicator test only to determine the general quality of the water. The sources of total dissolved solids can include all of the dissolved cations and anions, but the following table can be used as a generalization of the relationship of TDS to water quality problems.
Cations combined with Carbonates CaCO3, MgCO3 etc |
Associated with hardness, scale formation, bitter taste |
Cations combined with Chloride NaCl, KCl |
Salty or brackish taste, increase corrosivity |
An elevated total dissolved solids (TDS) concentration is not a health hazard. The TDS concentration is a secondary drinking water standard and, therefore, is regulated because it is more of an aesthetic rather than a health hazard. An elevated TDS indicates the following:
1) The concentration of the dissolved ions may cause the water to be corrosive, salty or brackish taste, result in scale formation, and interfere and decrease efficiency of hot water heaters; and
2) Many contain elevated levels of ions that are above the Primary or Secondary Drinking Water Standards, such as an elevated level of nitrate, arsenic, aluminum, copper, lead, etc.
Please find below links to BSS NanoMesh™ WPE ” Water Purification Equipment”


I was able to find good advice from your blog articles.